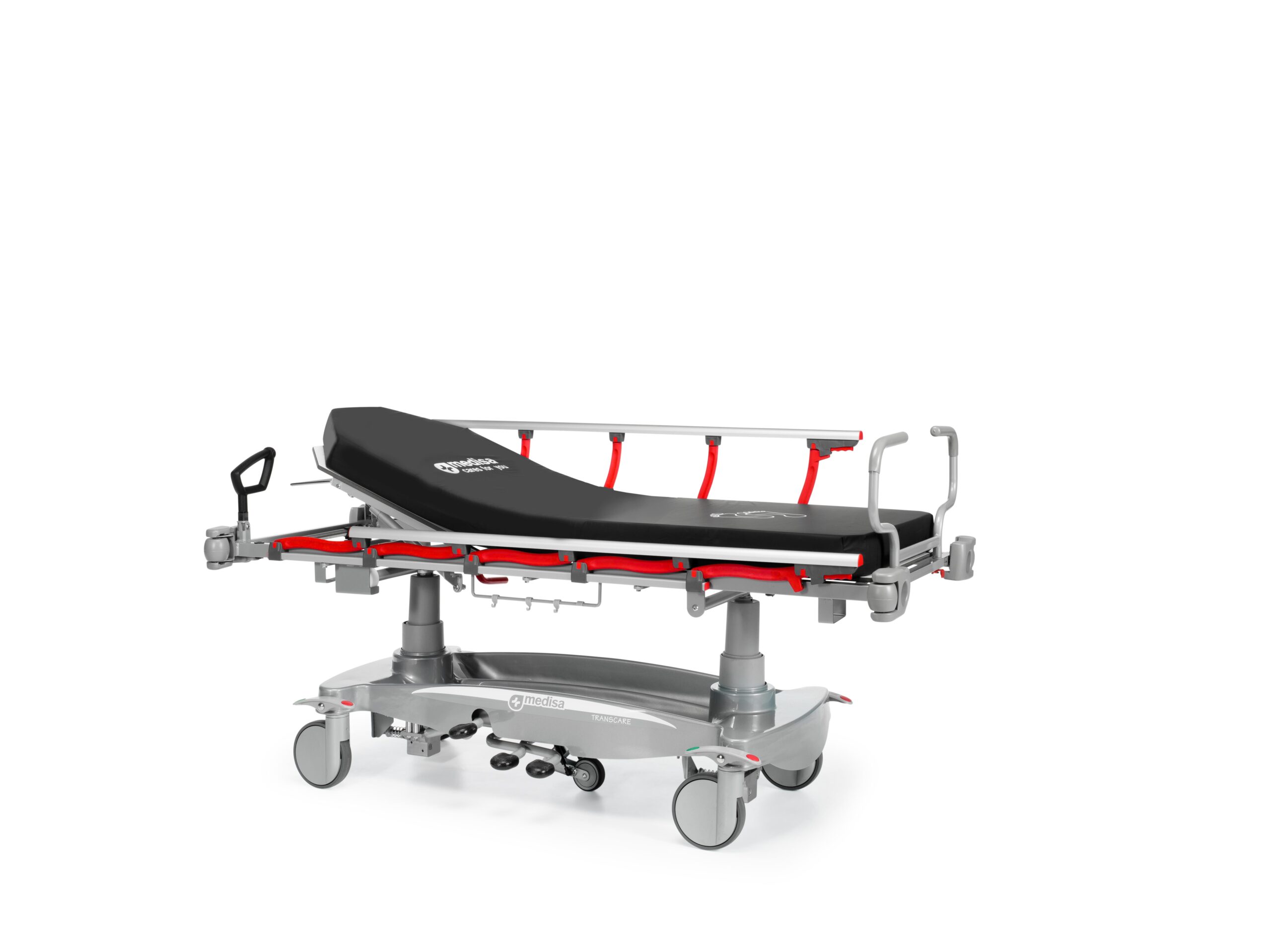
Characteristics
category
Mattress Base
Comfort
Handling
Safety
Structure made of steel tube with epoxy coating
Bed composed of 2 sections made of compact HPL material
Resistant to moisture and impact, allowing simple cleaning with a cloth with soapy water
Fully transparent radio mattress base
Useful area 100%
Support for additional IV poles (depending on size) in the corners
Urine bag hooks on each side of the stretcher
X-ray permeable mat, 10 cm thick
Padding made of high-density polyurethane foam, designed for optimal comfort and ergonomic suitability
Upholstered in self-extinguishing vinyl fabric
With four 200mm diameter, antistatic wheels and a wheel with servo steering
Centralized brake and directional lock via pedal located on each wheel
5th wheel diameter 125 mm servo assisted with gas spring
Folding handles to facilitate handling of the stretcher during transfer
Articulation of the trunk plane by pneumatic operation up to an angle of 90º
Height elevation from 515 mm to 820 mm using hydraulic columns
Trendelemburg / Reverse Trendelemburg ±16º by hydraulic drive
Bilateral pedal for hydraulic height adjustment and for Trendelenburg and anti-Trendelenburg control
Protective bumpers located in the corners
ABS lower fairing for easy cleaning, with built-in oxygen bottle holder
Folding side rails 150 long x 34 cm high; 29 cm distance between vertical bars
Easy and safe handling, without risk of bruising or entrapment
Its lowest position does not exceed the height of the mat
Technical file
Dimensions
- External dimension: 2040 x 890 mm
- Patient surface dimension: 1890 x 680 mm
- Backrest dimensions: 620 x 710 mm
- Legrest dimensions del plano de piernas: 660 x 1160 mm
Technical data
- Height adjustment: 515 – 820 mm
- Heigth adjustment with chassis support: 570 mm – 880 mm
- Backrest tilt: 90º
- Trendelenburg/Reverse Trendelenburg: ± 16º
- Maximum weight load: 317 kg
- Weight without accessories: 100 kg
Accessories
Included
Mat
Stainless steel IV Pole
Optional
Monitor holder support
Paper roll holder
Chassis holder and cassette holder support
Padding for railings
Transfer mat
Medical records support
Patient straps
Normative
All products manufactured by MEDISA comply with directive 2007/47/EEC on medical devices (Annex I and Annex VII).
All processes have been evaluated and certified according to the requirements of the UNE-EN ISO 9001:2008 standard for Quality Management and the UNE-EN ISO 14001:2004 standard for Environmental Management, and the UNE-EN ISO 13485 standard for medical devices.
MEDISA products are subject to regulations: IEC 60601-1, IEC 60601-1-2, IEC 60601-2-52
Certified by Bureau Veritas.
CERTIFICATES Nº ES047196-1 / ES047195-1
Scope: design, manufacturing, marketing and technical assistance of hospital beds and chairs. Marketing of hospital furniture.